 
Case report forms and serious event narratives.Case report form tabulations on the manufacturer’s clinical experience with the product.A description of the facility where the product is manufactured.A data summary of validation of important processes and assays involved in the manufacture of the product.Information on the manufacturing, chemistry, and controls of the product.Draft labeling of the product to be licensed.A clinical data section that includes safety and efficacy data on the product.Information on the applicant submitting the biologics license application.A summary of information submitted as part of the application.The Form FDA 356h contains the following information: However, as with New Drug Applications (NDA), an electronic Common Technical Document (eCTD) can be used to submit a biologics license application.Ī Form FDA 356h is an application to market a new drug, biologic, or antibiotic drug for use in humans. Next, applicants must submit a Form FDA 356h to the document control center at the Center for Biologics Evaluation and Research (CBER), a division of the FDA that specifically handles biologics.īoth paper and electronic submissions are acceptable. Read Also: What Makes for a Good Validation Master Plan? 2. In some cases, the manufacturer may also want to consider whether they want submit a validation plan for the FDA to review or to meet with the FDA regarding the validation plan before they submit the application. When determining the timing of the submission, they should consider the schedule and needs of the advisory committee. They also should schedule a bioresearch monitoring inspection. Applicants should identify a review committee and arrange a meeting with the Food and Drug Administration (FDA) before they submit the application. 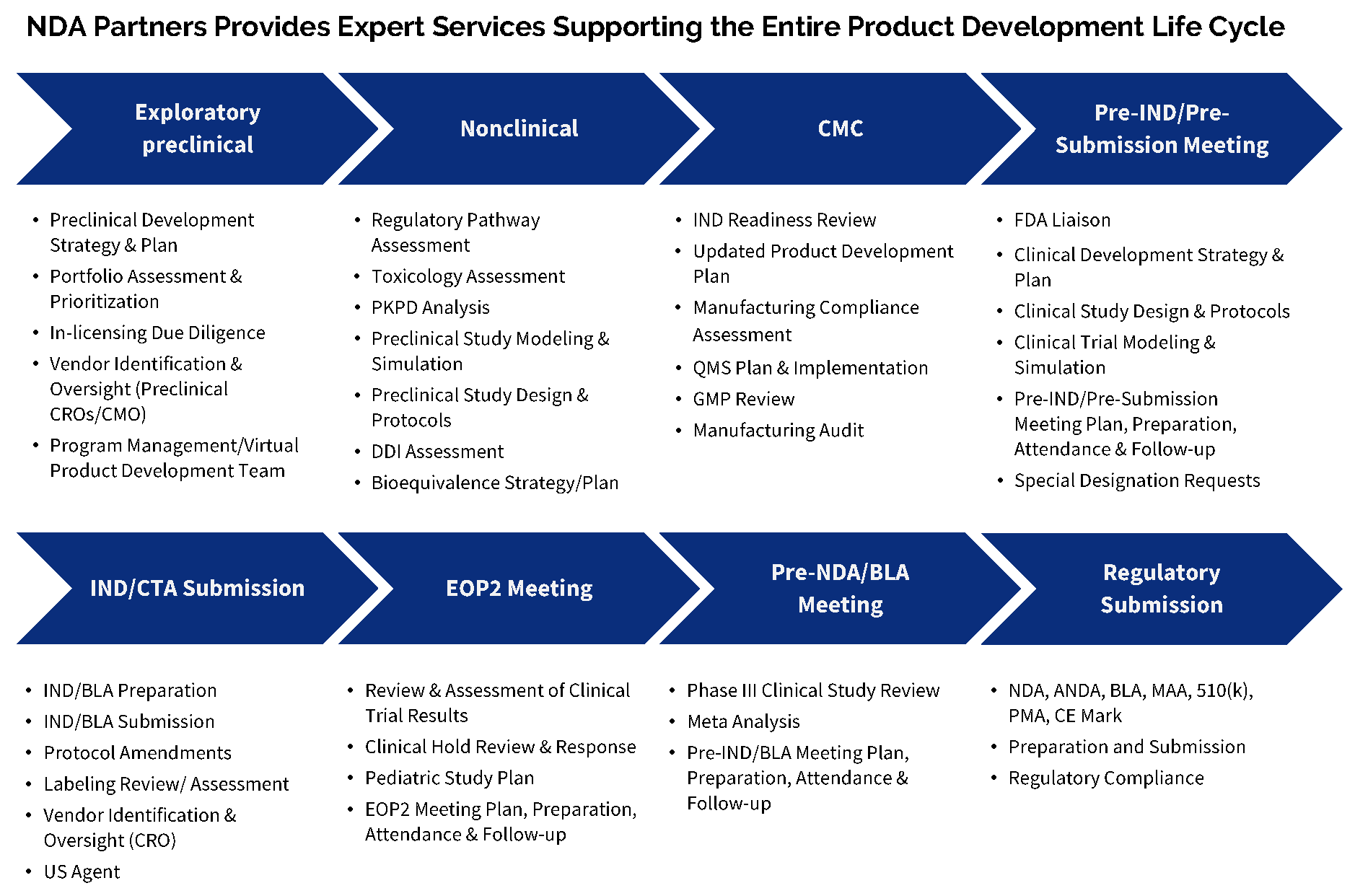
Apply for a Biologics License ApplicationĪnyone who is involved in the manufacture of biologics or who takes responsibility for the regulatory compliance of a biologic can apply for a biologics license application. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
